文章信息
- 马耀磊, 李霄, 李田甜, 等.
- MA Yaolei, LI Xiao, LI Tiantian, et al.
- 中医药调控线粒体畸变治疗射血分数保留型心力衰竭的研究进展
- Research progress in traditional Chinese medicine regulating mitochondrial aberration for the treatment of HFpEF
- 天津中医药, 2024, 41(11): 1475-1481
- Tianjin Journal of Traditional Chinese Medicine, 2024, 41(11): 1475-1481
- http://dx.doi.org/10.11656/j.issn.1672-1519.2024.11.19
-
文章历史
- 收稿日期: 2024-07-26
2. 天津中医药大学组分中药国家重点实验室, 天津 301617
心力衰竭(HF)是全球发病率和病死率最高的疾病之一[1],中国心血管疾病现患人数3.3亿,HF患者890万[2]。流行病学调查显示,射血分数保留型心力衰竭(HFpEF)患者数量达到HF患者总数的50%[3]。目前针对HFpEF的西医治疗包括减轻容量负荷、扩血管、抗炎等,这些治疗手段可以缓解HFpEF患者充血、水肿的症状并降低HF的住院风险[4-5]。但是这些治疗手段不能显著改善HFpEF患者的预后以及降低其病死率[5-6]。同时,HF患者长期应用扩血管制剂和利尿剂容易引起利尿剂抵抗、电解质紊乱等问题[7-8]。因此,研究HFpEF的潜在病理机制并确定新的治疗靶点变得至关重要。HFpEF的发展与能量平衡的破坏密切相关,HFpEF患者心脏腺嘌呤核苷三磷酸(ATP)水平只有正常人群心脏ATP水平的20%~40%[9]。这种现象出现的主要原因为线粒体畸变导致的线粒体结构和功能受损[10]。中药在治疗心血管疾病方面有着悠久的历史[11],多通过“益气温阳”治疗原则改善心肌能量代谢[12]。本文主要总结了线粒体畸变导致HFpEF的关键靶点,并针对上述靶点分析归纳出临床或临床前研究中能够潜在治疗HFpEF的中药,为中医药有效治疗HFpEF提供参考依据。
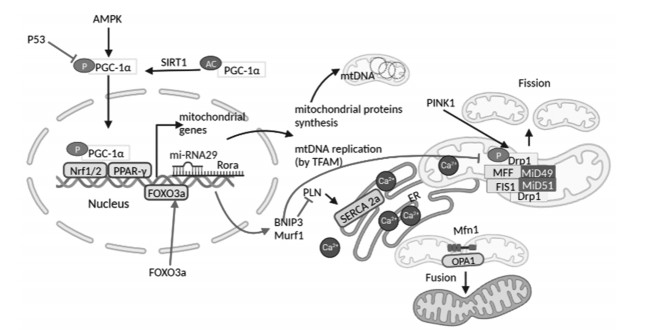
1 线粒体畸变诱发HFpEF的病理机制心脏是人体代谢最活跃的器官,其质量仅占体质量的0.5%,但却消耗了近8%的ATP。线粒体数量和功能是维持心脏代谢和保证心脏收缩舒张功能的基础[13]。临床研究表明,HFpEF患者会出现线粒体破裂、嵴破坏和线粒体面积减少等现象[10]。而线粒体畸变是导致线粒体结构、功能异常的主要原因。故调控线粒体畸变并维持线粒体的结构和功能正常对于HFpEF患者是至关重要的[14],其中线粒体畸变包括线粒体的生物发生障碍、线粒体动力学障碍和线粒体自噬异常。见图 1。
|
| 注:P53,人体抑癌基因;AMPK,AMP依赖的蛋白激酶;PGC-1α,过氧化物酶体增殖物激活受体γ-辅激活因子1-α;SIRT1,沉默信息调节因子1;Nrf1/2,核呼吸因子1/2;PPAR-γ,过氧化物酶体增殖物激活受体;FOXO3a,叉形头转录因子的O亚型3;Rora,RAR相关孤儿受体α;mtDNA,线粒体DNA;TFAM,线粒体转录因子A;BNIP3,BCL2相互作用蛋白3;Murf1,肌肉特异性环指蛋白1;SERCA 2a,肌浆网/内质网Ca2+ ATP酶2a;PINK1,磷酸酶及张力蛋白同源物(PTEN)诱导的激酶1;Drp1,动力蛋白相关蛋白1;OPA1,视神经萎缩蛋白1;MFF,线粒体分裂因子;FIS1,线粒体裂变蛋白1;MiD49/51,49kDa和51kDa线粒体动力学蛋白。 图 1 线粒体畸变诱发HFpEF的病理机制 Fig. 1 Pathological mechanism of mitochondrial aberration-induced HFpEF |
线粒体生物发生是从已有线粒体再生以维持线粒体功能的过程。线粒体生物发生障碍导致线粒体数量减少和ATP生成不足,促进HF的发展。线粒体生物发生由过氧化物酶体增殖物激活受体γ辅助活化因子1(PGC1)与其他转录因子结合驱动[15]。HF患者[16]、HF动物模型中[17]PGC1以及磷酸化PGC1的表达均减弱,这是线粒体生物发生异常的主要原因。最新研究表明,HFpEF在老年人群中发病率较高,这可能与短端粒过度激活p53(tumor protein p53)导致的PGC1抑制相关[18-20]。
1.1.1 PGC1PGC1作为核编码转录调节因子能够激活线粒体DNA(mtDNA)转录,进而驱动线粒体生物发生。在小鼠中过表达PGC1α会导致心脏线粒体的数量和体积显著增加,这与线粒体生物发生相关基因的上调相吻合[21-22]。动物实验和人体试验表明,在HF模型中PGC1α的表达和活性降低[23-24]。PGC1的活性受到PGC1磷酸化、乙酰化以及表达含量的调控。沉默信息调节因子1(SIRT1)作为烟酰胺腺嘌呤二核苷酸(NAD+)依赖性脱乙酰化酶能够去乙酰化PGC1,非乙酰化的PGC1转录活性进一步增强[25]。此外,作为丝氨酸/苏氨酸激酶的AMP依赖蛋白激酶(AMPK)在AMP/ATP比率升高时被激活。其中,ATP生成减少导致AMPK活化并发生磷酸化从而激活PGC1,磷酸化的PGC1转位到细胞核中与核呼吸因子-1(NRF-1)相互作用,并促进线粒体转录因子A(TFAM)的表达。同时,TFAM能够增强线粒体功能以弥补HF所致的能量受损状态[25-26]。PGC1的缺失可导致线粒体基因表达受到抑制、线粒体结构紊乱、线粒体酶活性异常和ATP产生减少,从而加速心功能障碍[27-30]。PGC1α的激活能够恢复线粒体功能和超微结构,进一步明显改善心脏舒张功能并缓解HFpEF患者存在的线粒体异常状态[31]。
1.1.2 P53P53作为心脏转录调节因子,参与协调心脏结构、激活兴奋-收缩偶联、影响线粒体生物发生和促进氧化磷酸化相关蛋白转录等功能。P53缺陷导致PGC1α/β表达增加、mtRNA合成增加、TFAM转录增强[32],进而促进线粒体生物发生和影响线粒体功能。衰老所致的短端粒能够激活P53,并且驱动线粒体发生障碍,这是老年人易发生HFpEF的潜在机制[18]。高脂饮食(HFD)联合L-NAME[33]以及端粒酶RNA组分TERC(telomerase RNA)敲除的HFpEF小鼠模型中,衰老降低了P53激活的阈值。P53持续激活导致PGC1α和PGC1β受到抑制,进而使得TFAM、mtDNA介导的三羧酸循环(TCA)、β-氧化关键酶转录抑制[34-35]。上述原因均会导致线粒体生物发生减少,使线粒体表现出嵴缺失和面积减少等现象,从而影响心肌舒缩功能并驱动HFpEF发生发展的进程[19]。特异性敲除心肌细胞中P53的动物不能发生HFpEF[19],因此抑制P53表达以恢复线粒体生物发生是治疗HFpEF的潜在途径。
1.2 线粒体裂变/融合动力学障碍线粒体是不断经历裂变和融合的动态结构,线粒体融合形成细长的相互连接的线粒体网络,裂变产生离散的碎片线粒体以维持线粒体和心脏功能[36]。线粒体的融合裂变可以消除DNA受损的线粒体并保证mtDNA的完整性,这一过程主要由线粒体裂变蛋白动力蛋白相关蛋白(DRP1)、线粒体分裂蛋白(Fis1)和融合蛋白线粒体融合蛋白(MFN)、OPA1(Optic Atrophy 1)调节[37-38]。线粒体融合和裂变的失衡导致线粒体过度融合,并且使受损线粒体不能及时清除而诱导HFpEF的发生。
1.2.1 DRP1线粒体裂变由DRP相关的GTP酶及其线粒体表面受体调控[38]。DRP1和Fis1是线粒体裂变的关键蛋白[39]。当线粒体发生裂变时,DRP1从细胞质募集到线粒体裂变部位,进而与Fis1、线粒体裂变因子(MFF)和线粒体动力学蛋白相互作用,促进线粒裂变[40-41]。调控DRP1活性可用于调节线粒体动力学和功能,其中包括线粒体呼吸、ATP产生和线粒体膜电位等[42-43]。在高血压诱导HFpEF的大鼠模型中,DRP1 Ser616磷酸化水平降低,从而导致线粒体裂变受损以及线粒体功能障碍[44]。DRP1 Ser616磷酸化由丝氨酸/苏氨酸蛋白激酶(PTEN)诱导激酶1(PINK1)介导。PINK1在心肌特异性过表达增强了DRP1的线粒体定位和Ser616磷酸化激活,进一步促进了ATP产生和线粒体裂变增加。线粒体裂变增加能去除过度融合的受损线粒体,并延缓高血压诱导的HFpEF的进展[45]。然而,DRP1 Ser637磷酸化会抑制线粒体分裂。活化的FOXO3a易位进入细胞核激活BNIP3,活化的BNIP3阻断DRP1 Ser637磷酸化。过表达FOXO3a使线粒体过度裂变和碎片化,促使HFpEF发生不良进展[46]。因此靶向DRP1蛋白和DRP1的翻译后修饰(PTM)是HFpEF潜在的治疗方法。
1.2.2 MFN1/2线粒体融合通过交换线粒体内容物(代谢物、蛋白、mtDNA)来恢复受损线粒体活性,维持线粒体代谢效率。MFN1/2和OPA1是线粒体融合所必需的蛋白[47]。MFN缺乏导致会引起线粒体融合速率降低和线粒体形态异常[48]。PGC1α与雌激素相关受体(ERR)结合能够靶向MFN启动子并且激活MFN蛋白表达[49]。MFN在线粒体之间形成同型寡聚体(MFN1-MFN1或MFN2-MFN2)和异质寡聚体(MFN1-MFN2)复合体[50],介导线粒体外膜发生融合。OPA1依赖线粒体膜电位介导线粒体内膜的融合。同时,线粒体内膜融合能够维持线粒体嵴的形成以及mtDNA生成[50-51]。在小鼠实验中,OPA1、MFN2抑制会导致线粒体形态和细胞结构发生变化[52-53],从而导致严重的心脏扰动和HF,增加OPA1的表达能够减轻心肌缺血再灌注损伤[54]。以上结果表明,促进线粒体融合能够缓解心脏损伤[55]。在压力过载(POL)诱导的HFpEF模型中存在明显的线粒体融合、裂变以及平衡紊乱,但是MFN、OPA1蛋白表达与射血分数降低型心力衰竭(HFrEF)组、正常组并没有显著差异[14]。上述实验结果表明,MFN1/2和OPA1作为线粒体融合的关键蛋白可能为治疗HFpEF提供新的靶点,但是这一途径仍需更多的相关研究来证实。
1.3 线粒体自噬异常线粒体自噬能够去除损伤严重且无法通过融合修复的线粒体,从而维持线粒体质量控制过程和细胞线粒体库整体健康[56]。线粒体生物发生、融合、裂变旨在维持稳定的线粒体生物功能。如果上述方式无法挽救受损的线粒体,那么受损的线粒体必须通过线粒体自噬进行清除。这是维持线粒体稳态的重要过程。但是,这种适应性线粒体自噬的持续激活会引起线粒体过度自噬,进而损伤线粒体和心肌细胞功能[57]。线粒体自噬通过受体介导的FUNDC1/BNIP3和非受体介导的PINK1/Parkin通路进行调节。在心肌细胞中,AMPK诱导的PINK1自噬激活能够延缓HF进展并减轻心脏损伤[58]。而在高脂诱导的心脏损伤模型中,PINK1介导自噬的激活会加重心脏损伤[59]。心肌细胞BNIP3过表达表现出适应性自噬激活后去除受损线粒体并保护心脏功能[60],而压力超负荷的HF模型中,BNIP3激活介导的线粒体自噬促进了HF发展[61],因此,研究HFpEF疾病进展中不同靶点介导线粒体自噬激活的程度、强度和持续时间可能为HFpEF治疗提供有针对性的措施。
心肌细胞线粒体自噬由PINK1和E3泛素连接酶Parkin介导。在线粒体膜电位降低的情况下,线粒体膜激酶PINK1发生磷酸化并激活胞质Parkin。Parkin激活后移位进入线粒体,进而介导线粒体外膜蛋白的泛素化。线粒体外膜蛋白泛素化传递自噬信号并促进受损线粒体向溶酶体的递送和吞噬[62]。晚期HF患者的PINK1蛋白水平会显著降低。小鼠PINK1的缺失以及主动脉弓缩窄术(TAC)诱导的HF模型中,PINK1和线粒体Parkin蛋白水平降低,这与线粒体自噬活性的变化一致。PINK1表达降低导致心脏线粒体功能障碍、早期心室功能障碍和病理性心脏肥大[63-65]。HFpEF患者存在明显的脂肪酸氧化供能不足和心肌细胞脂肪酸堆积等现象,长期脂毒性会加重线粒体损伤。此时由PINK1/Parkin介导的线粒体自噬被抑制,受损的线粒体无法降解,线粒体出现形态改变和异质性,最终导致心肌细胞损伤、心脏收缩和舒张功能障碍[66]。因此,激活PINK1以清除受损线粒体是干预HFpEF的有效措施。
2 调控线粒体畸变治疗HFpEF的中药 2.1 中药复方HFpEF的病机主要是气虚血瘀,而补阳还五汤作为益气活血方能够显著改善HFpEF[67]。在腹主动脉缩窄致舒张性HF模型中,补阳还五汤抑制线粒体超微机构肿胀、空泡变形并促进线粒体数量增加。补阳还五汤还能抑制Drp1 mRNA表达,抑制线粒体过度分裂,保护线粒体结构和功能,维持舒张性HF时正常心肌能量代谢、减缓HF进程[68]。
气阴两虚HFpEF患者应用生脉散进行辨证治疗具有一定安全性且心功能和中医症状得到改善[69]。生脉散增强了沉默信息调节因子1(SIRT1) 和p-AMPKα蛋白水平,并降低了PGC-1α乙酰化水平,在糖尿病所致舒张功能障碍的心肌病小鼠模型中能够缓解线粒体功能障碍[70]。
芪苈强心胶囊是首个进入《舒张性心力衰竭诊断和治疗专家共识》的中成药[71]。在醋酸脱氧皮质酮(DOCA)-盐敏感性高血压HFpEF大鼠模型中,芪苈强心胶囊可以显著增加线粒体SERCA蛋白表达,改善舒张期Ca2+增加导致的心肌舒张异常[72]。在自发性高血压大鼠(SHR)舒张功能障碍模型中,芪苈强心胶囊通过上调AMPK/PGC-1α轴,保留了线粒体含量和ATP的产生,进而改善SHR模型中的左心室舒张功能障碍和心脏重塑[73]。
苓桂术甘汤是HFpEF患者指南推荐用药,能够显著改善HFpEF患者临床症状[74]。苓桂术甘汤改善了盐敏感(Dahl/SS)大鼠HFpEF模型中心肌线粒体的损伤,增加心肌ATP含量。并且苓桂术甘汤上调HFpEF模型心肌组织的SIRT3、PGC-1α、PPARa和p-AMPK表达。显著缓解HFpEF大鼠的左心室结构重塑和舒张功能障碍[75]。
2.2 中药有效成分黄芪是HF患者常用中药之一。黄芪甲苷作为黄芪的主要成分和质量标志物,在HFD联合L-NAME致小鼠HFpEF模型中,黄芪甲苷能够改善HFpEF小鼠心肌线粒体肿胀和嵴结构的紊乱[76]。
虎杖具有利湿退黄,清热解毒,散瘀止痛,止咳化痰的功能。白藜芦醇是虎杖水溶性部位中的苯酚类化合物,具有抗氧化、抗炎、治疗心血管疾病等多种药理作用。在Dahl/SS大鼠模型中,白藜芦醇处理倾向于保留线粒体质量和生物发生,并恢复了MFN1和OPA1蛋白表达,使得线粒体聚变/裂变保持平衡[77],改善了高血压HF大鼠的线粒体功能障碍。以上研究提示白藜芦醇可能是影响线粒体功能从而治疗HFpEF的潜力单体化合物。调控线粒体畸变治疗HFPEF的中药总结见表 1。

|
心脏舒张功能受损是HFpEF的典型特征,目前尚无针对HFpEF的有效治疗方法,而HFpEF的患病人数仍在持续增长,高居不下的病死率和致残率带来了巨大的社会医疗负担。心肌线粒体畸变导致的能量代谢异常是HFpEF心脏舒张功能受损的主要诱因,也是HFpEF治疗的重点,总结HFpEF中线粒体畸变的病理机制为提出有针对性的治疗方案提供了理论依据。本研究总结出PGC1、P53、MFN1/2、OPA1、PINK1等靶点是参与调控HFpEF患者心肌线粒体畸变的靶标蛋白。针对上述机制已有相应小分子药物进入了临床研究,但是单一靶点的治疗措施容易产生许多不良反应且预后较差。传统中药以多途径、多通路、多靶点发挥治疗作用,这也契合了HFpEF中心脏能量代谢紊乱的复杂机制。黄芪、人参、丹参等传统活血化瘀、补气类中药在HF的治疗中已经拥有长久的临床应用和循证证据,本研究总结了能够有效调控心肌能量代谢和改善线粒体功能的中药,有望为HFpEF的中药治疗提供更多证据支持。
| [1] |
HEIDENREICH P A, BOZKURT B, AGUILAR D, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: executive summary: a report of the American college of cardiology/American heart association joint committee on clinical practice guidelines[J]. Circulation, 2022, 145(18): e876-e894. |
| [2] |
中国心血管健康与疾病报告2022概要[J]. 中国循环杂志, 2023, 38(6): 583-612.
|
| [3] |
LIU J, GUO Z J, FAN M D, et al. Major depression and clinical outcomes in patients with heart failure with preserved ejection fraction[J]. European Journal of Clinical Investigation, 2021, 51(3): e13401. DOI:10.1111/eci.13401 |
| [4] |
周京敏, 王华, 黎励文. 射血分数保留的心力衰竭诊断与治疗中国专家共识2023[J]. 中国循环杂志, 2023, 38(4): 375-393. DOI:10.3969/j.issn.1000-3614.2023.04.001 |
| [5] |
MCDONAGH T A, METRA M, ADAMO M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure[J]. European Heart Journal, 2021, 42(36): 3599-3726. DOI:10.1093/eurheartj/ehab368 |
| [6] |
Correction to: 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American college of cardiology/American heart association joint committee on clinical practice guidelines[J]. Circulation, 2023, 147(14): e674.
|
| [7] |
WANG Y, WANG Q Y, LI C, et al. A review of Chinese herbal medicine for the treatment of chronic heart failure[J]. Current Pharmaceutical Design, 2017, 23(34): 5115-5124. |
| [8] |
闫少华. 沙库巴曲缬沙坦对射血分数保留心力衰竭患者影响的Meta分析[D]. 承德: 承德医学院, 2023.
|
| [9] |
CAPONE F, SOTOMAYOR-FLORES C, BODE D, et al. Cardiac metabolism in HFpEF: from fuel to signalling[J]. Cardiovascular Research, 2023, 118(18): 3556-3575. DOI:10.1093/cvr/cvac166 |
| [10] |
DE JONG K A, LOPASCHUK G D. Complex energy metabolic changes in heart failure with preserved ejection fraction and heart failure with reduced ejection fraction[J]. Canadian Journal of Cardiology, 2017, 33(7): 860-871. DOI:10.1016/j.cjca.2017.03.009 |
| [11] |
FU S F, ZHANG J H, GAO X M, et al. Clinical practice of traditional Chinese medicines for chronic heart failure[J]. Heart Asia, 2010, 2(1): 24-27. DOI:10.1136/ha.2009.001123 |
| [12] |
陈绍熠, 金艳蓉, 柴琳, 等. 慢性心衰心肌能量代谢中医研究进展[J]. 中国民族民间医药, 2022, 31(13): 55-58, 84. DOI:10.3969/j.issn.1007-8517.2022.13.zgmzmjyyzz202213014 |
| [13] |
BROWN D A, PERRY J B, ALLEN M E, et al. Expert consensus document: mitochondrial function as a therapeutic target in heart failure[J]. Nature Reviews Cardiology, 2017, 14(4): 238-250. DOI:10.1038/nrcardio.2016.203 |
| [14] |
CHAANINE A H, JOYCE L D, STULAK J M, et al. Mitochondrial morphology, dynamics, and function in human pressure overload or ischemic heart disease with preserved or reduced ejection fraction[J]. Circulation Heart Failure, 2019, 12(2): e005131. DOI:10.1161/CIRCHEARTFAILURE.118.005131 |
| [15] |
QIU Z L, WEI Y, SONG Q Q, et al. The role of myocardial mitochondrial quality control in heart failure[J]. Frontiers in Pharmacology, 2019, 10: 1404. DOI:10.3389/fphar.2019.01404 |
| [16] |
KARAMANLIDIS G, NASCIMBEN L, COUPER G S, et al. Defective DNA replication impairs mitochondrial biogenesis in human failing hearts[J]. Circulation Research, 2010, 106(9): 1541-1548. DOI:10.1161/CIRCRESAHA.109.212753 |
| [17] |
RIEHLE C, WENDE A R, ZAHA V G, et al. PGC-1β deficiency accelerates the transition to heart failure in pressure overload hypertrophy[J]. Circulation Research, 2011, 109(7): 783-793. DOI:10.1161/CIRCRESAHA.111.243964 |
| [18] |
SAHIN E, COLLA S, LIESA M, et al. Telomere dysfunction induces metabolic and mitochondrial compromise[J]. Nature, 2011, 470(7334): 359-365. DOI:10.1038/nature09787 |
| [19] |
CHEN X N, LIN H, XIONG W Y, et al. p53-dependent mitochondrial compensation in heart failure with preserved ejection fraction[J]. Journal of the American Heart Association, 2022, 11(11): e024582. DOI:10.1161/JAHA.121.024582 |
| [20] |
CHANG A C, ONG S G, LAGORY E L, et al. Telomere shortening and metabolic compromise underlie dystrophic cardiomyopathy[J]. Proceedings of the National Academy of Sciences of the United States of America, 2016, 113(46): 13120-13125. |
| [21] |
RUSSELL L K, MANSFIELD C M, LEHMAN J J, et al. Cardiac-specific induction of the transcriptional coactivator peroxisome proliferator-activated receptor gamma coactivator-1alpha promotes mitochondrial biogenesis and reversible cardiomyopathy in a developmental stage-dependent manner[J]. Circulation Research, 2004, 94(4): 525-533. DOI:10.1161/01.RES.0000117088.36577.EB |
| [22] |
LEHMAN J J, BARGER P M, KOVACS A, et al. Peroxisome proliferator-activated receptor gamma coactivator-1 promotes cardiac mitochondrial biogenesis[J]. Journal of Clinical Investigation, 2000, 106(7): 847-856. DOI:10.1172/JCI10268 |
| [23] |
BRAINARD R E, FACUNDO H T. Cardiac hypertrophy drives PGC-1α suppression associated with enhanced O-glycosylation[J]. Biochimica et Biophysica Acta(BBA)-Molecular Basis of Disease, 2021, 1867(5): 166080. DOI:10.1016/j.bbadis.2021.166080 |
| [24] |
PACKER M. Longevity genes, cardiac ageing, and the pathogenesis of cardiomyopathy: implications for understanding the effects of current and future treatments for heart failure[J]. European Heart Journal, 2020, 41(39): 3856-3861. DOI:10.1093/eurheartj/ehaa360 |
| [25] |
RODGERS J T, LERIN C, HAAS W, et al. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1[J]. Nature, 2005, 434(7029): 113-118. DOI:10.1038/nature03354 |
| [26] |
TIAN L, CAO W J, YUE R J, et al. Pretreatment with Tilianin improves mitochondrial energy metabolism and oxidative stress in rats with myocardial ischemia/reperfusion injury via AMPK/SIRT1/PGC-1 alpha signaling pathway[J]. Journal of Pharmacological Sciences, 2019, 139(4): 352-360. DOI:10.1016/j.jphs.2019.02.008 |
| [27] |
MARTIN O J, LAI L, SOUNDARAPANDIAN M M, et al. A role for peroxisome proliferator-activated receptor γ coactivator-1 in the control of mitochondrial dynamics during postnatal cardiac growth[J]. Circulation Research, 2014, 114(4): 626-636. DOI:10.1161/CIRCRESAHA.114.302562 |
| [28] |
HUSS J M, IMAHASHI K, DUFOUR C R, et al. The nuclear receptor ERRalpha is required for the bioenergetic and functional adaptation to cardiac pressure overload[J]. Cell Metabolism, 2007, 6(1): 25-37. DOI:10.1016/j.cmet.2007.06.005 |
| [29] |
KÄRKKÄINEN O, TUOMAINEN T, MUTIKAINEN M, et al. Heart specific PGC-1α deletion identifies metabolome of cardiac restricted metabolic heart failure[J]. Cardiovascular Research, 2019, 115(1): 107-118. DOI:10.1093/cvr/cvy155 |
| [30] |
LI Y, FENG Y F, LIU X T, et al. Songorine promotes cardiac mitochondrial biogenesis via Nrf2 induction during sepsis[J]. Redox Biology, 2021, 38: 101771. DOI:10.1016/j.redox.2020.101771 |
| [31] |
HUANG S Y, CHEN X N, PAN J N, et al. Hydrogen sulfide alleviates heart failure with preserved ejection fraction in mice by targeting mitochondrial abnormalities via PGC-1α[J]. Nitric Oxide, 2023, 136: 12-23. |
| [32] |
MAK T W, HAUCK L, GROTHE D, et al. p53 regulates the cardiac transcriptome[J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114(9): 2331-2336. |
| [33] |
SCHIATTARELLA G G, ALTAMIRANO F, TONG D, et al. Nitrosative stress drives heart failure with preserved ejection fraction[J]. Nature, 2019, 568(7752): 351-356. DOI:10.1038/s41586-019-1100-z |
| [34] |
OHTSUJI M, KATSUOKA F, KOBAYASHI A, et al. Nrf1 and Nrf2 play distinct roles in activation of antioxidant response element-dependent genes[J]. Journal of Biological Chemistry, 2008, 283(48): 33554-33562. DOI:10.1074/jbc.M804597200 |
| [35] |
NANJAIAH H, VALLIKANNAN B. Lutein upregulates the PGC-1α, NRF1, and TFAM expression by AMPK activation and downregulates ROS to maintain mtDNA integrity and mitochondrial biogenesis in hyperglycemic ARPE-19 cells and rat retina[J]. Biotechnology and Applied Biochemistry, 2019, 66(6): 999-1009. DOI:10.1002/bab.1821 |
| [36] |
GOTTLIEB R A, BERNSTEIN D. Mitochondrial remodeling: rearranging, recycling, and reprogramming[J]. Cell Calcium, 2016, 60(2): 88-101. DOI:10.1016/j.ceca.2016.04.006 |
| [37] |
SKULACHEV V P. Mitochondrial filaments and clusters as intracellular power-transmitting cables[J]. Trends in Biochemical Sciences, 2001, 26(1): 23-29. DOI:10.1016/S0968-0004(00)01735-7 |
| [38] |
WESTERMANN B. Mitochondrial fusion and fission in cell life and death[J]. Nature Reviews Molecular Cell Biology, 2010, 11(12): 872-884. DOI:10.1038/nrm3013 |
| [39] |
LIN J J, CHEN K, CHEN W F, et al. Paradoxical mitophagy regulation by PINK1 and TUFm[J]. Molecular Cell, 2020, 80(4): 607-620. DOI:10.1016/j.molcel.2020.10.007 |
| [40] |
BOUTRY M, KIM P K. ORP1L mediated PI(4)P signaling at ER-lysosome-mitochondrion three-way contact contributes to mitochondrial division[J]. Nature Communications, 2021, 12(1): 5354. DOI:10.1038/s41467-021-25621-4 |
| [41] |
KALKHORAN S B, KRISTON-VIZI J, HERNANDEZ-RESENDIZ S, et al. Hydralazine protects the heart against acute ischaemia/reperfusion injury by inhibiting Drp1-mediated mitochondrial fission[J]. Cardiovascular Research, 2022, 118(1): 282-294. DOI:10.1093/cvr/cvaa343 |
| [42] |
SCHMITT K, GRIMM A, DALLMANN R, et al. Circadian control of DRP1 activity regulates mitochondrial dynamics and bioenergetics[J]. Cell Metabolism, 2018, 27(3): 657-666. DOI:10.1016/j.cmet.2018.01.011 |
| [43] |
CHO H M, RYU J R, JO Y, et al. Drp1-Zip1 interaction regulates mitochondrial quality surveillance system[J]. Molecular Cell, 2019, 73(2): 364-376. DOI:10.1016/j.molcel.2018.11.009 |
| [44] |
WU S N, LU Q L, WANG Q L, et al. Binding of FUN14 domain containing 1 with inositol 1, 4, 5-trisphosphate receptor in mitochondria-associated endoplasmic reticulum membranes maintains mitochondrial dynamics and function in hearts in vivo[J]. Circulation, 2017, 136(23): 2248-2266. DOI:10.1161/CIRCULATIONAHA.117.030235 |
| [45] |
SHOU J, HUO Y L. PINK1 phosphorylates Drp1S616 to improve mitochondrial fission and inhibit the progression of hypertension-induced HFpEF[J]. International Journal of Molecular Sciences, 2022, 23(19): 11934. DOI:10.3390/ijms231911934 |
| [46] |
CHAANINE A H, KOHLBRENNER E, GAMB S I, et al. FOXO3a regulates BNIP3 and modulates mitochondrial calcium, dynamics, and function in cardiac stress[J]. American Journal of Physiology Heart and Circulatory Physiology, 2016, 311(6): H1540-H1559. DOI:10.1152/ajpheart.00549.2016 |
| [47] |
CHEN H, MCCAFFERY J M, CHAN D C. Mitochondrial fusion protects against neurodegeneration in the cerebellum[J]. Cell, 2007, 130(3): 548-562. DOI:10.1016/j.cell.2007.06.026 |
| [48] |
CHEN H, DETMER S A, EWALD A J, et al. Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development[J]. 2003, 160(2): 189-200.
|
| [49] |
LIESA M, BORDA-D'AGUA B, MEDINA-GÓMEZ G, et al. Mitochondrial fusion is increased by the nuclear coactivator PGC-1beta[J]. PLoS One, 2008, 3(10): e3613. DOI:10.1371/journal.pone.0003613 |
| [50] |
CHANDHOK G, LAZAROU M, NEUMANN B. Structure, function, and regulation of mitofusin-2 in health and disease[J]. Biological Reviews of the Cambridge Philosophical Society, 2018, 93(2): 933-949. DOI:10.1111/brv.12378 |
| [51] |
COGLIATI S, FREZZA C, SORIANO M E, et al. Mitochondrial cristae shape determines respiratory chain super complexes assembly and respiratory efficiency[J]. Cell, 2013, 155(1): 160-171. DOI:10.1016/j.cell.2013.08.032 |
| [52] |
SABBAH H N, GUPTA R C, SINGH-GUPTA V, et al. Abnormalities of mitochondrial dynamics in the failing heart: normalization following long-term therapy with elamipretide[J]. Cardiovascular Drugs and Therapy, 2018, 32(4): 319-328. DOI:10.1007/s10557-018-6805-y |
| [53] |
CHEN L, GONG Q Z, STICE J P, et al. Mitochondrial OPA1, apoptosis, and heart failure[J]. Cardiovascular Research, 2009, 84(1): 91-99. DOI:10.1093/cvr/cvp181 |
| [54] |
WANG M, WANG R Y, ZHOU J H, et al. Calenduloside E ameliorates myocardial ischemia-reperfusion injury through regulation of AMPK and mitochondrial OPA1[J]. Oxidative Medicine and Cellular Longevity, 2020, 2020: 2415269. |
| [55] |
JAVADOV S, RAJAPUROHITAM V, KILI A, et al. Expression of mitochondrial fusion-fission proteins during post-infarction remodeling: The effect of NHE-1 inhibition[J]. Basic Research in Cardiology, 2011, 106(1): 99-109. DOI:10.1007/s00395-010-0122-3 |
| [56] |
SHIRES S E, GUSTAFSSON Å B. Regulating renewable energy: Connecting AMPKα2 to PINK1/parkin-mediated mitophagy in the heart[J]. Circulation Research, 2018, 122(5): 649-651. DOI:10.1161/CIRCRESAHA.118.312655 |
| [57] |
HOSHINO A, MITA Y, OKAWA Y, et al. Cytosolic p53 inhibits Parkin-mediated mitophagy and promotes mitochondrial dysfunction in the mouse heart[J]. Nature Communications, 2013, 4: 2308. DOI:10.1038/ncomms3308 |
| [58] |
ZHU P J, WAN K, YIN M, et al. RIPK3 induces cardiomyocyte necroptosis via inhibition of AMPK-parkin-mitophagy in cardiac remodelling after myocardial infarction[J]. Oxidative Medicine and Cellular Longevity, 2021, 2021: 6635955. DOI:10.1155/2021/6635955 |
| [59] |
WANG S H, ZHU X L, WANG F, et al. LncRNA H19 governs mitophagy and restores mitochondrial respiration in the heart through Pink1/Parkin signaling during obesity[J]. Cell Death & Disease, 2021, 12(6): 557. |
| [60] |
ZHANG J, NEY P A. Role of BNIP3 and NIX in cell death, autophagy, and mitophagy[J]. Cell Death and Differentiation, 2009, 16(7): 939-946. DOI:10.1038/cdd.2009.16 |
| [61] |
CHAANINE A H, JEONG D, LIANG L, et al. JNK modulates FOXO3a for the expression of the mitochondrial death and mitophagy marker BNIP3 in pathological hypertrophy and in heart failure[J]. Cell Death & Disease, 2012, 3(2): 265. |
| [62] |
WANG B, NIE J L, WU L J, et al. AMPKα2 protects against the development of heart failure by enhancing mitophagy via PINK1 phosphorylation[J]. Circulation Research, 2018, 122(5): 712-729. DOI:10.1161/CIRCRESAHA.117.312317 |
| [63] |
FRATI G, VECCHIONE C, SCIARRETTA S. Novel beneficial cardiovascular effects of natural activators of autophagy[J]. Circulation Research, 2018, 123(8): 947-949. DOI:10.1161/CIRCRESAHA.118.313530 |
| [64] |
SHIRAKABE A, ZHAI P Y, IKEDA Y, et al. Drp1-dependent mitochondrial autophagy plays a protective role against pressure overload-induced mitochondrial dysfunction and heart failure[J]. Circulation, 2016, 133(13): 1249-1263. DOI:10.1161/CIRCULATIONAHA.115.020502 |
| [65] |
WU C N, ZHANG Z, ZHANG W D, et al. Mitochondrial dysfunction and mitochondrial therapies in heart failure[J]. Pharmacological Research, 2022, 175: 106038. DOI:10.1016/j.phrs.2021.106038 |
| [66] |
SHAO D, KOLWICZ S C Jr, WANG P, et al. Increasing fatty acid oxidation prevents high-fat diet-induced cardiomyopathy through regulating parkin-mediated mitophagy[J]. Circulation, 2020, 142(10): 983-997. DOI:10.1161/CIRCULATIONAHA.119.043319 |
| [67] |
吴甜. 补阳还五汤合防己黄芪汤治疗射血分数保留型心力衰竭气虚血瘀水停证的疗效观察[D]. 长沙: 湖南中医药大学, 2023.
|
| [68] |
王臻, 李洁白, 董昕, 等. 补阳还五汤对舒张性心衰大鼠心肌α-MHC、β-MHC、Drp1 mRNA及线粒体超微结构的影响[J]. 北京中医药大学学报, 2018, 41(12): 1002-1006. DOI:10.3969/j.issn.1006-2157.2018.12.006 |
| [69] |
原思敏. 生脉散合桂枝甘草龙骨牡蛎汤治疗老年射血分数保留型心衰的临床疗效观察[D]. 南京: 南京中医药大学, 2021.
|
| [70] |
TIAN J, TANG W Z, XU M, et al. Shengmai San alleviates diabetic cardiomyopathy through improvement of mitochondrial lipid metabolic disorder[J]. Cellular Physiology and Biochemistry, 2018, 50(5): 1726-1739. DOI:10.1159/000494791 |
| [71] |
廖玉华, 杨杰孚, 张健, 等. 舒张性心力衰竭诊断和治疗专家共识[J]. 临床心血管病杂志, 2020, 36(1): 1-10. |
| [72] |
郝佳梦, 常丽萍, 王璐, 等. 基于网络药理学及实验验证探究芪苈强心胶囊治疗射血分数保留型心衰的作用机制[J]. 中草药, 2022, 53(14): 4365-4375. |
| [73] |
WANG J F, LI Z M, WANG Y Y, et al. Qiliqiangxin enhances cardiac glucose metabolism and improves diastolic function in spontaneously hypertensive rats[J]. Evidence-Based Complementary and Alternative Medicine, 2017, 2017: 3197320. DOI:10.1155/2017/3197320 |
| [74] |
慢性心力衰竭中医诊疗指南(2022年)[J]. 中医杂志, 2023, 64(7): 743-756.
|
| [75] |
刘东敏. 苓桂术甘汤对射血分数保留心衰线粒体功能及SIRT3信号通路的作用机制研究[D]. 成都: 成都中医药大学, 2022.
|
| [76] |
王笑. 黄芪甲苷改善射血分数保留型心力衰竭的药效作用机制研究[D]. 天津: 天津中医药大学, 2023.
|
| [77] |
RIMBAUD S, RUIZ M, PIQUEREAU J, et al. Resveratrol improves survival, hemodynamics and energetics in a rat model of hypertension leading to heart failure[J]. PLoS One, 2011, 6(10): e26391. DOI:10.1371/journal.pone.0026391 |
2. State Key Laboratory of Component-based Chinese Medicine, Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China
 2024, Vol. 41
2024, Vol. 41